Iloprost (Ventavis®)
|
Indications: Iloprost is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve a composite endpoint consisting of exercise tolerance, symptoms (NYHA Class), and lack of deterioration.
Mechanism: Iloprost is a synthetic analog of prostacyclin PGI2. Iloprost dilates systemic and pulmonary arterial vascular beds. It also affects platelet aggregation but the relevance of this effect to the treatment of pulmonary hypertension is unknown.
Dosing: Iloprost is intended to be inhaled using the I-neb® AAD® System. Patients should receive 6 to 9 doses (inhalations) per day (minimum of 2 hours between doses during waking hours) as follows:
- Starting dose: 2.5 mcg. Up-titrate to 5 mcg if 2.5 mcg is well tolerated.
- Maintenance dose: 5 mcg.
- Iloprost should be taken 6 to 9 times per day (no more than once every 2 hours) during waking hours, according to individual need and tolerability. The maximum daily dose evaluated in clinical studies was 45 mcg (5 mcg 9 times per day).
Efficacy: A randomized, double-blind, multi-center, placebo-controlled trial was conducted in 203 adult patients (inhaled iloprost: n=101; placebo: n=102) with NYHA Class III or IV pulmonary arterial hypertension. Patients received 2.5 or 5.0 mcg of iloprost by repeated inhalations 6 to 9 times per day during waking hours, in addition to patients’ current therapy.
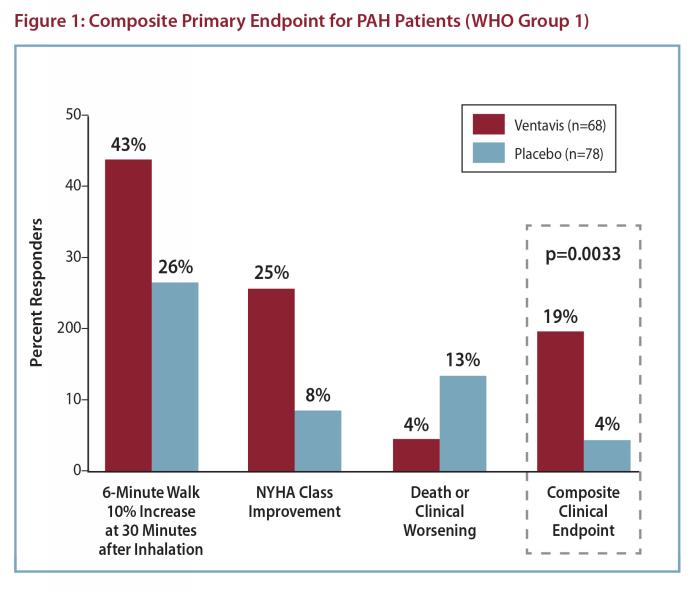
The primary efficacy endpoint was clinical response at 12 weeks, a composite endpoint defined by:
a) improvement in exercise ability (6-minute walk test) by at least 10% versus baseline evaluated 30 minutes after dosing, b) improvement by at least one NYHA class versus baseline, and c) no death or deterioration
of pulmonary hypertension. Although effectiveness was seen in the full population (response rates for the primary composite endpoint of 17% and 5%; p=0.007), there was inadequate evidence of benefit in patients with pulmonary hypertension associated with chronic thromboembolic disease (WHO Group 4); the results presented are therefore those related to patients with PAH (WHO Group 1). The response rate for the primary efficacy endpoint among PAH patients was 19% for the iloprost group, compared with 4% for the placebo group (p=0.0033). All three components of the composite endpoint favored iloprost (Figure 1).
|
|
|
|
|
|
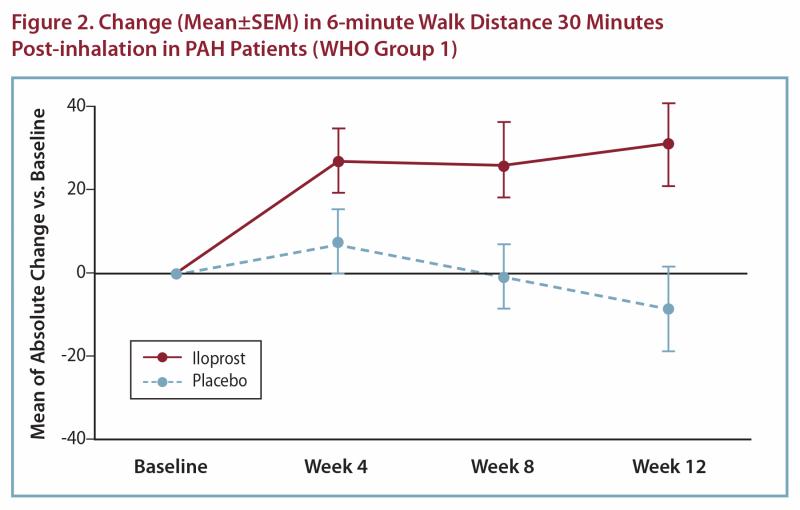
The absolute change in 6-minute walk distance (Figure 2) measured 30 minutes after inhalation among patients with PAH was greater in the iloprost group compared to the placebo group at all time points. At Week 12, the placebo-corrected difference was 40 meters (p<0.01). When walk distance was measured immediately prior to inhalation, the improvement compared to placebo was approximately 60% of the effect seen at 30 minutes after inhalation.
|
|
|
|
|
|
Adverse Events:
Most common (>3% placebo-adjusted) adverse reactions are vasodilation (flushing), cough increased, headache, trismus, insomnia, nausea, hypotension, vomiting, alkaline phosphatase increased, flu syndrome, back pain, tongue pain, palpitations, syncope, GGT increased, muscle cramps, hemoptysis, and pneumonia
Warnings:
- Hypotension leading to syncope has been observed. Iloprost should not be administered in patients with systolic blood pressure below 85 mmHg.
- Pulmonary venous hypertension: Discontinue if pulmonary edema is present.
- May cause bronchospasm: Patients with a history of hyper-reactive airway disease may be more sensitive.
Contraindications: none
Metabolism/Drug interactions: Iloprost has the potential to increase the hypotensive effect of vasodilators and antihypertensive agents. There is a potential for increased risk of bleeding, particularly in patients maintained on anticoagulants.
Access Program: Upon prescribing Ventavis, patients are automatically enrolled in the PAH Pathways program. They are contacted by a patient case manager and the specialty pharmacy nurses in order to handle reimbursement and Ventavis dosage and administration training.
Patient Enrollment form: http://www.4ventavis.com/pdf/Ventavis_Patient_Enrollment_Forms.pdf
Full US Prescribing Information can be found here: http://www.4ventavis.com/pdf/Ventavis_PI.pdf
|